TECHNICAL PAPER # 32
UNDERSTANDING WATER SUPPLY
AND TREATMENT FOR INDIVIDUAL
AND SMALL COMMUNITY SYSTEMS
By Stephen A. Hubbs
Technical Reviewers
Dr. F. O. Blackwell
Paul S. Fardig
Morton S. Hilbert
VITA
1600
Wilson Boulevard, Suite 500
Arlington, Virginia 22209 USA
Tel: 703/276-1800 * Fax:
703/243-1865
Internet: pr-info@vita.org
Understanding Water Supply & Treatment
for
Individual & Small Community Systems
ISBN: 0-86619-240-9
[C] 1985,
Volunters in Technical Assistance
PREFACE
This paper is one of a series published by Volunteers in
Technical
Assistance to provide an introduction to specific
state-of-the-art
technologies of interest to people in developing countries.
The papers are intended to be used as guidelines to help
people choose technologies that are suitable to their
situations.
They are not intended to provide construction or
implementation
details. People are
urged to contact VITA or a similar organization
for further information and technical assistance if they
find that a particular technology seems to meet their needs.
The papers in the series were written, reviewed, and
illustrated
almost entirely by VITA Volunteer technical experts on a
purely
voluntary basis.
Some 500 volunteers were involved in the production
of the first 100 titles issued, contributing approximately
5,000 hours of their time.
VITA staff included Maria Giannuzzi
as editor, Suzanne Brooks handling typesetting and layout,
and
Margaret Crouch as project manager.
The author of this paper, VITA Volunteer Stephen A. Hubbs,
is an
environmental engineer with the Louisville Water Company in
Louisville,
Kentucky. He has
worked with the World Health Organization
in Switzerland, Germany, and Holland.
The reviewers are also
VITA volunteers. Dr.
F. O. Blackwell is an associate professor of
environmental health with the East Carolina University
School of
Allied Health. He
has worked as a health and sanitation advisor
in Pakistan, and has taught at the American University of
Beirut, Lebanon, School of Public Health.
He is a registered
professional engineer and has worked in the field of
environmental
health in 20 countries in Africa, South America, Central
America, and Asia.
Paul S. Fardig specializes in environmental
health and sanitation, with a focus on water supply and
sewage
disposal for small towns and villages, including basic
sanitary
engineering. Morton
S. Hilbert is Professor and Chairman of the
Department of Environmental and Industrial Health at the
University
of Michigan.
VITA is a private, nonprofit organization that supports
people
working on technical problems in developing countries.
VITA offers
information and assistance aimed at helping individuals and
groups to select and implement technologies appropriate to
their
situations. VITA
maintains an international Inquiry Service, a
specialized documentation center, and a computerized roster of
volunteer technical consultants; manages long-term field
projects;
and publishes a variety of technical manuals and papers.
UNDERSTANDING
WATER SUPPLY AND TREATMENT FOR INDIVIDUAL
AND SMALL COMMUNITY SYSTEMS
by
VITA Volunteer Stephen A. Hubbs
I. INTRODUCTION
The design, construction, and operation of small-scale water
treatment systems for individual homes and small communities
represent a significant challenge to public health because
of the
wide variety of water quality conditions in developing
countries.
Because developing countries often lack expertise for
designing
and operating such systems, these systems are often
developed
under extreme limitations of both materials and
personnel. For
this reason, any system considered for individual homes or
small
communities in developing countries must achieve the basic
goals
of water purification through simple design, operation, and
maintenance.
For water to be considered suitable for drinking, it should
be
aesthetically pleasing; that is, it should look, smell, and
taste
good. It must also
be wholesome; that is, it should not contain
any substances that cause sickness or disease
(pathogens). These
two characteristics are mutually important in that water must
be
"acceptable" to consumers before they will use it,
and free of
harmful agents if it is to be used safely.
It is not uncommon
for consumers to select water that is aesthetically pleasing
but
of questionable wholesomeness, over less aesthetically pleasing
water that is free of disease agents.
Consumers tend to judge the
quality of water by the way it looks and tastes, rather than
also
taking into account the wholesomeness of the water.
The ideal small-scale water treatment system would be
affordable,
simple to design, construct, and operate; and capable of
changing
unacceptable water to water that is free of taste, odor,
turbidity
(cloudiness or discoloration), and disease agents in a
single
process. Another
desirable feature would be for the system to
stop operating automatically if it is producing water that
is not
fit for consumption; that is, it should operate only if it
is
operating properly.
In reality, however, there is no perfect
system.
Nevertheless, in developing a system, the designer
should always strive to achieve adequate quantity in the
least
technically complicated way.
This paper provides guidelines on how to choose a water
source,
and how to purify and retrieve water to ensure that it is
safe
for human consumption.
Applications are general in nature, relying
on the creativity of the system designer to draw from
whatever resources are available to develop a water
treatment
system capable of improving the water supply.
II. BASIC THEORY OF WATER SUPPLY
THE HYDROLOGIC CYCLE
The hydrologic cycle (water cycle) traces the path of water
from
the oceans to the atmosphere, rivers, ground, swamps, and
eventually
back to the oceans (Figure 1).
As the water progresses
37p03.gif (600x600)
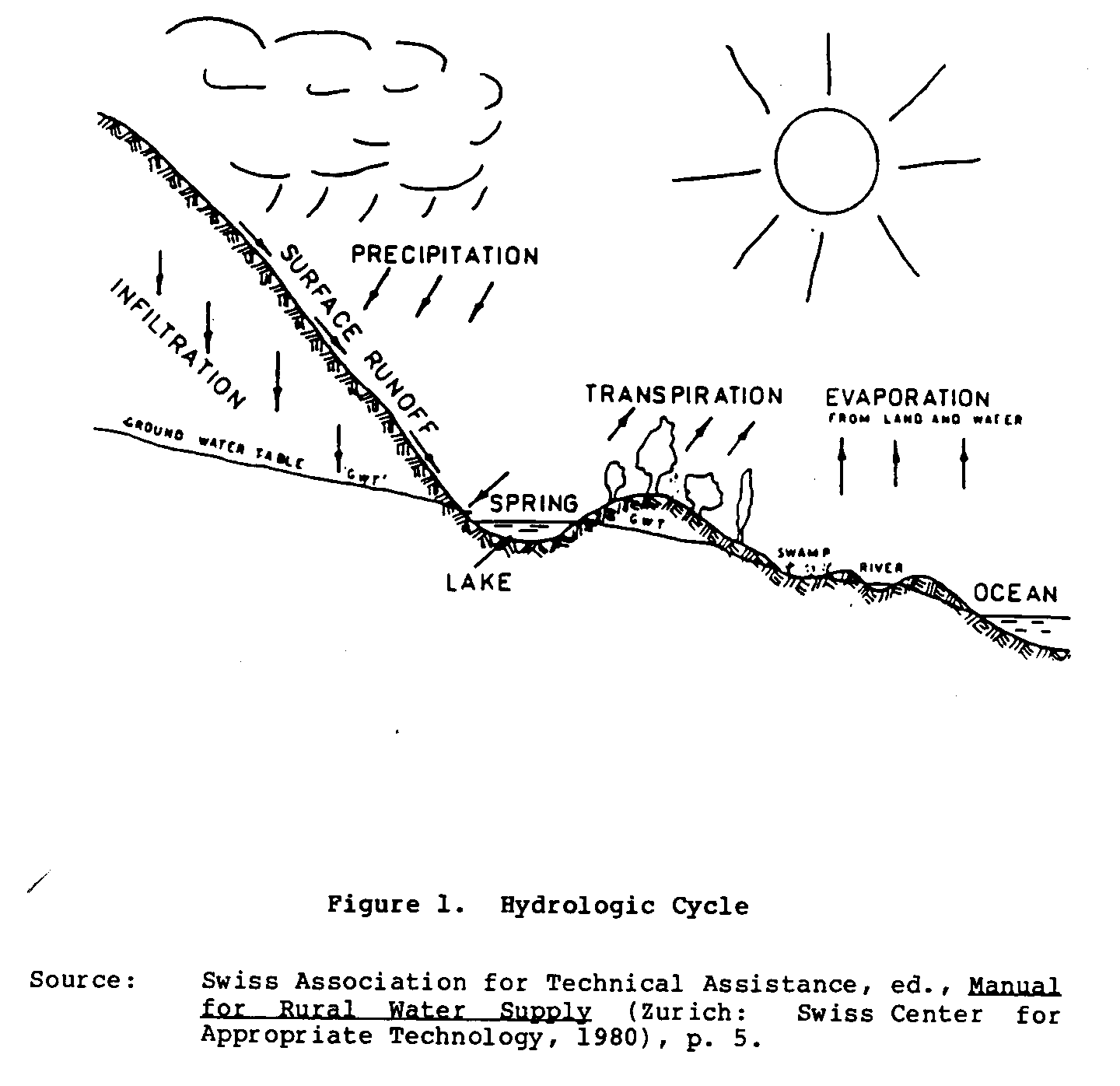
through the various stages of the hydrologic cycle, it is
affected
by many factors that determine its ultimate quality.
The water
can be extracted for use at any stage in the cycle; however,
the
quantity and quality of water available often limits the
user to
only a few choices.
For drinking water, it is important to
select a water source that provides an adequate supply of
water
of the highest quality possible.
SOURCES OF WATER
Precipitation
In areas where air pollution is not a major factor, rain
water
can provide a suitable, high-quality source of water.
Typically,
rain is collected from rooftops through gutters and stored
in
tanks or cisterns (underground storage vessels).
Because the
roof (or any collection surface) is subject to contamination
from
nesting and flying birds and airborne dust, one cannot
assume
that this source of water is suitable for consumption.
Underground
storage chambers are subject to infiltration as well as
leakage. Problems
with infiltration can be serious, as water
from nearby outdoor toilets and subsurface sewage disposal
systems
can enter the cistern when the water level in the cistern is
low. For these
reasons, rain water must always be disinfected
before it is consumed.
Periodic inspection of the cistern is
recommended, with annual cleaning to remove any sediment
that has
accumulated.
The cistern should be sized to provide an adequate supply of
water throughout low rainfall seasons.
In many situations, this
will limit the feasibility of using rain water as a
year-round
source of water. The
amount of water available is easily
calculated by multiplying the annual or seasonal average
rainfall
(in meters) by the surface area of the collecting surface
(in
square meters).
Provisions for screening out large particles
(leaves) and keeping out small animals should be included in
any
storage system.
Springs
A spring represents a point in the hydrologic cycle where
ground
water meets the land surface and flows into a stream.
The water
quality at the point of surfacing is often excellent, as the
water has usually traveled, or percolated, through thick
layers
of soil. In this
process of percolation, the water picks up
dissolved minerals (calcium, magnesium, iron, etc.) and is
purified
of biological pathogens (disease producing organisms).
The
spring will exhibit varying quantity and quality depending
on the
geologic formation in the area.
A continuously flowing spring
that is always clear can provide a good source of drinking
water.
In selecting springs as a source of supply, particular
caution
should be used in areas of what is called Karst (limestone)
topography. These
areas typically contain many sinkholes, or
depressions, through which surface drainage is transported
to the
ground water (Figure 2).
Water entering the ground water by this
37p05.gif (600x600)
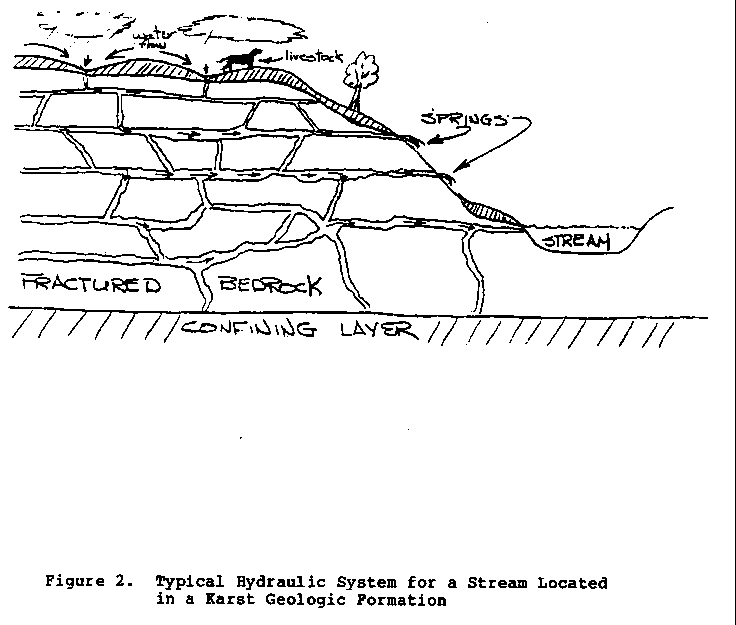
path bypasses the percolation process that purifies it.
As a
result, springs in these areas can produce poor-quality
water
much like surface water, and must be treated appropriately.
Ground Water
If a stream is located in a sand and gravel stratum, a
supply of
suitable drinking water might be obtained easily by drilling
or
digging a well into the aquifer that feeds the river (Figure
3).
37p06.gif (600x600)
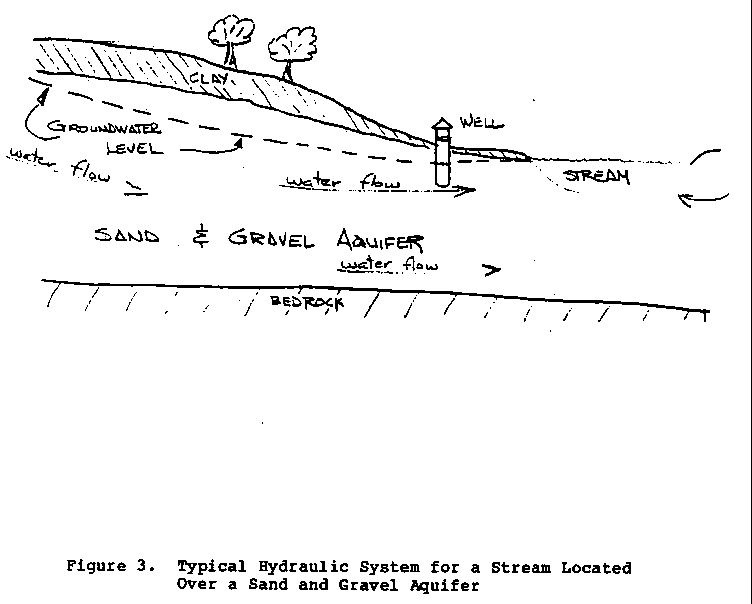
Since surface streams typically define the lowest hydraulic
gradient
in an area, a well dug into the sand and gravel will
typically
draw water from highland areas; if these areas have not
undergone extensive development or become contaminated (such
as
by dumps and landfills), they will usually provide sanitary
water. As with
spring water, ground water from limestone strata
must be suspect quality.
Depending on local geological conditions,
however, the water may contain unacceptably high levels of
iron, manganese, and/or salt, making it unpalatable.
Ground water can be extracted from any point in the geologic
formation, but the depth and type of cover over the ground
water
will determine the feasibility of constructing a well for
water
supply. Water from a
well typically exhibits a constant quality.
When the well is properly constructed to eliminate surface
contamination,
it can provide an excellent source of drinking water.
Surface Streams
Villages are typically established near a source of water
and
transportation routes, as these two factors often determine
their
habitability and reason for existence.
The source of water for
villages is typically surface water.
Surface water can be used
to supply water for drinking and washing; it can be a means
of
transportation; it can be used for irrigation, livestock
watering,
or for sewage disposal.
These multiple uses are often
conflicting, and the water source may not be able to meet
all the
demands placed upon it.
While surface streams, rivers, and lakes often represent the
most
accessible supply of water to a village, they are also the
most
vulnerable to contamination.
Surface water typically has highly
variable water quality, and can be the source of many
diseases.
To be suitable for consumption, surface water must always be
treated to remove harmful substances.
WATER SOURCE SELECTION
For either surface water supply or ground water supply, the
point
of water withdrawal should be made as far upstream as
possible.
Two principal drawbacks to this concept are (1) people
living
below the water source must travel greater distances to
obtain
their water; and (2) the higher the source of water, the
less
volume of water there is.
A basic understanding of the topography
and geology of the area can help in locating the best
point of water withdrawal.
In selecting a source of water, attention should be given to
the
use of land in the immediate watershed and the chance of
contamination
to the source.
Problems with unreliable water quality
can be largely eliminated or reduced by avoiding areas that
will
likely be contaminated by human waste water,
agricultural/livestock
runoff, and industrial discharge.
The most important step
in developing a potable water supply is the selection of the
QRhighest-quality water source possible.
It is difficult to define categorically a particular source
of
water as superior to another.
However, ground water supplies and
rain water do have a greater chance of being free of serious
contamination than do surface water supplies.
Of the surface
supplies, springs that provide clear water under all
conditions
and that are located in areas that do not have numerous
sinkholes
are preferred over surface streams.
Any surface water, including
clear-running mountain streams, can be contaminated by
pathogens
and must be treated before use.
No matter what source of water
is being considered, the local factors influencing the water
quality must always be evaluated.
If possible, one should call
upon the local health authorities to analyze the suitability
of a
particular water source.
WATER EXTRACTION AND TRANSPORT
There are many ways of extracting and transporting water
from a
source to the point of use.
Water can be taken from streams and
wells by hand and transported in buckets or ceramic vessels.
Where materials and technology are available, water can be
pumped
by electric, diesel, or wind-powered pumps and transported
through pipelines. In
situations where the source is located at
an altitude higher than the point of use, the water can be
transported by gravity.
A detailed discussion of these techniques
exceeds the scope of this paper; to obtain this information,
readers are directed to other VITA publications.
Caution should be used in determining how the water will be
extracted and transported.
Extreme care should be exercised to
avoid contamination of the water.
Whenever possible, hand powered
or machine-powered pumps should be installed, and the use of
buckets, which can contaminate the source, avoided.
Pumps also
allow a well to be sealed, eliminating the possibility of
foreign
objects or contaminated surface water getting into the well.
WATER TREATMENT
This section discusses relatively simple, reliable, and
efficient
methods of treating water to remove solids and
pathogens. Methods
for the removal of additional toxic compounds (e.g., heavy
metals, industrial solvents, pesticides) are beyond the
scope of
this paper and are not covered here.
Water treatment for any fresh-water system basically
involves the
removal of solids, the removal of pathogens (disease-causing
bacteria, viruses, and other microbials), and the removal of
substances that impart bad tastes and odors.
In isolated instances,
additional toxic compounds must be removed before the
water can be drunk.
In supplying water to individual homes and
villages in rural areas, it is therefore far more desirable
to
locate a water source free of such toxic agents, because the
removal of such agents can be technically difficult and
economically
burdensome.
Solids in water may be of no health concern in
themselves. However,
solids (clay, organic material, etc.) in water can protect
pathogens from disinfection, and result in water quality
problems
even in treated systems.
Turbid drinking water is not particularly
appealing, which may lead consumers to select an alternate
source of clear water.
In doing so, however, unknowing consumers
could end up drinking water that is not wholesome, even
though it
appears to be of higher quality.
Thus, one goal in the treatment
of water should be the removal of suspended solids.
Solids in water can be divided into three categories:
those that
float, those that sink, and those that are suspended (that
is,
they neither float nor sink within reasonable periods of
time).
Of these three categories, the suspended solids are the most
difficult to remove.
Floating solids can be avoided by drawing
water from below the surface of the water source.
Solids that
settle without chemical treatment can often be removed by
allowing
the water to remain for one day or more in a facility
designed
for quiescent conditions (low water velocities).
Suspended
solids, however, must be removed by either chemical or
physical treatment methods.
To remove them in this way involves
more sophisticated equipment and a higher level of
maintenance.
Sedimentation
Sedimentation; or removal of those solids that sink, was
commonly
the only treatment provided to turbid streams through the
1800s.
This process relies on the rate at which the material in the
water settles or sinks, and the retention of water in such a
manner as to allow the material to reach the bottom of the
basin.
In sedimentation basins, it is important to remember that
the
principal design variable is the surface area of the basin,
not
the overall volume.
The basin need only be deep enough to ensure
good hydraulic flow patterns.
Proper design of inlet and outlet
structures is necessary to prevent the system from
short-circuiting,
and to avoid the removal of deposits from the floor of the
basin.
Sedimentation rates for solids can vary from 10 meters/hour
for
heavy silts to less than 0.005 meters/hour (5 mm/hour) for
fine
clays. Thus, the
composition of the solids in the water will
determine the feasibility and design criteria for the
sedimentation
process. Fine clay
suspensions and water with high color
content can be treated chemically to make the particles
settle
more readily. Such
treatment, called chemical coagulation, requires
the availability of chemicals, chemical feed equipment,
and routine sludge removal for proper operation.
Aluminum and
iron salts (alum, ferric sulfate) are typically used when
available,
along with organic polymers.
Maintaining these processes
is expensive and requires trained personnel.
Thus, chemical
coagulation is not typically considered for
individual/village
water supplies.
A sedimentation basin can be made of any suitable
material. It
can be as simple as a clay pot or as complicated as a
concrete
basin with continuous sludge drawoff.
Consideration should be
given to the amount of solids that will be collected in the
basin, and the methods of solids removal that will be
used. If
the solids are to be removed in a batch operation (requiring
the
temporary halting of the operation), additional units will
be
necessary if a continuous supply of water is required.
In general,
the additional units should be provided if possible,
although
this can cause an increase in overall construction costs.
The dimensions of a particular basin are determined by the
particles
to be settled, land constraints, the need for long-term
storage, and other physical and economic conditions.
Technical
assistance in designing the facility should be sought
whenever
available.
Storing water for extended periods of time can result in the
destruction of bacteria, as well as turbidity removal.
Storage
for two weeks or longer can remove up to 90 percent of
disease-causing
organisms. This process,
however, is not effective for
removing all pathogenics, and fine turbidity will remain in
suspension. In
addition, algae may grow in the water during this
time, making the water taste and smell bad.
In general, water
storage is a beneficial pretreatment if algae growth is not
a
problem. Caution
must be taken so far as possible to prevent the
contamination of the storage area by human and animal
wastes.
Filtration
Filtration has long been recognized as an effective method
of
water purification. The
ancient Egyptians recognized that boiling
and filtering (among other less proven techniques) were
capable of rendering foul water suitable for drinking.
Prior to
1700, it was commonly believed that filtration could remove
salt
from sea water. In
the 18th and 19th centuries, many patents
were issued in France and England for various filtration
devices,
both small units for in-house use and larger filters for
municipalities.
These filters used sand, cinders, charcoal, sponge,
wool, and many other materials.
The earliest mention of the mode
of action in slow sand filters was in the 1840s when an
Englishman
noted in a chemistry text that the filter media served to
support "finer materials of mud or
precipitate...which...form the
bed that really filters water."
This citation recognizes the
importance of the formation of a filtering layer that must
be
allowed to develop on top of the sand before the filter can
operate efficiently.
Slow sand filters (so named because of the relatively slow
downward
speed or velocity maintained in the filters) have been noted
as being effective for solids removal and bacterial
reduction
for over two centuries.
These early filters were not effective
for highly turbid streams, however, because of the short
filter
runs experienced before clogging.
The processes of chemical
coagulation and sedimentation paved the way for the
development
of rapid sand filters, which became popular in the early
1900s.
A few modern treatment plants still use slow sand
filtration,
although the standard for most large utilities is chemical
coagulation,
sedimentation (although direct filtration is becoming
increasingly popular), and rapid filtration through mixed
media.
This paper is limited to slow sand filtration only, because
it
requires simple operating conditions and generally produces
high-quality
water. Adequate
units range from sand-filled drums or
earthen-lined basins to concrete structures with complex
under-drain
systems. Each type
of unit suits a particular situation.
A simple filter, designed for domestic use, can be made from
a
55-gallon drum and sand.
It can improve the quality of surface
water significantly, as long as initial turbidities are not
too
high. As with any
slow filter, the surface of the filter must be
kept wet to maintain the biological growth known as the
"schmutzdecke."
(The schmutzdecke consists of a variety of biologically
active microorganisms that break down organic matter,
while much of the suspended inorganic matter is retained by
straining). This
type of filter can produce 10 to 20 liters of
water per hour if operated continuously, but intermittent
operation
is more typical. In
such an operation, the flow rate
through the filter should be limited so as not to exceed
optimum
rates (10 to 20 liters per hour).
The filter should be kept
covered to eliminate algae growth and contamination from
dust.
For proper filtration, the surface of the filter should
always be
kept submerged.
The selection of materials for the construction of a
domestic
filter will depend mainly on what resources are
available. If a
55-gallon drum is selected, the interior of the drum must be
protected against rusting.
Containers that have been used for
storing pesticides, herbicides, and other toxic chemicals
must
not be used. The
preferred filter medium is sand with an effective
size in the range of 0.15 to 0.35 mm.
Ungraded river sand is
acceptable if nothing else is available.
The sand should be
thoroughly washed by panning to remove very fine sand,
clays, and
organic matter. The
sand should be placed in the container in a
layer about 1 meter deep, and arranged with inlet-outlet
piping
to allow easy operation.
A typical slow sand filter is shown in
Figure 4.
37p13.gif (486x486)
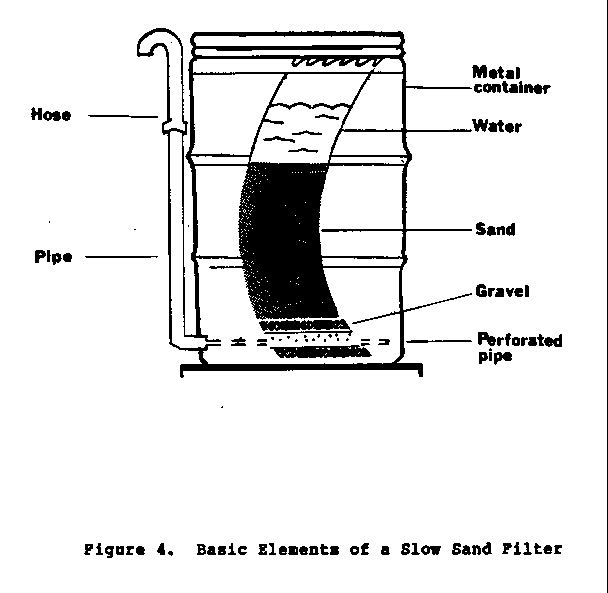
The design and operation of a slow sand filter for a small
village
should be supervised by a qualified person.
The design
criteria should take into account available materials and
funds,
as well as the suitability of the water source for
filtration.
Regardless of the efficiency of a sand filter for removing
turbidity
and reducing bacteria, sand filters alone should not be
considered adequate for the treatment of contaminated
surface
waters. In every
case, some form of disinfection should also be
used if the water is to be used for human consumption.
Disinfection
Although sedimentation and filtration can greatly reduce the
amount of bacteria in contaminated water, the reliability of
these two processes to produce water suitable for drinking
is
limited. Many
pathogens can survive even after these processes
are operated properly.
Removal of pathogens can be almost negligible
when the processes are carried out improperly.
It is
necessary that any water from a contaminated source be
disinfected
before consumption, if at all possible.
Disinfection can be accomplished by mechanical, chemical,
and
thermal techniques.
(Other techniques, such as radiation, are
beyond the scope of this paper.)
If the water is sufficiently
free of suspended solids, it can be passed through a
small-pore
filter, which is capable of physically blocking the path of
microorganisms.
Certain stone filters have this capability, but
the filtering rate is relatively slow.
Chemical agents, particularly
the halogens (chlorine, bromine, iodine), have been
demonstrated
to be highly efficient in killing bacteria.
A universally
recognized method for killing bacteria is boiling, which can
destroy life forms in even turbid suspensions.
Each method of
disinfection has its limitations, which should be recognized
before the technology is adopted.
A recent evaluation of ceramic filters which are capable of
meeting WHO standards for bacterial quality indicated that
of all
the strainers tested, only carved stone filters were capable
of
yielding acceptable bacterial quality by straining
alone. Other
filters, impregnated with silver, were effective, but the
mode of
disinfection was not limited to straining alone.
The carved
stone filter was effective, but it was also relatively heavy
and
expensive. It should
be noted that filters that strain out the
test organisms (coliform bacteria) do not necessarily also
remove
the pathogenic viruses, which are typically much smaller
than
bacteria. One should
be cautious, therefore, in interpreting the
results of straining for pathogen removal based upon
indicator
organisms.
The ability of filtration and straining to remove large
numbers
of pathogens should be emphasized.
Properly filtered water is
considered to be far more healthful than unfiltered
water. However,
the complete removal of pathogens cannot be guaranteed.
For this reason, water must undergo further disinfection
through
chlorination or boiling.
These two disinfection methods are
discussed in the sections that follow.
Chlorination
Chemical agents such as chlorine, bromine, and iodine have
been
used to eliminate waterborne diseases in major water
supplies
since the early 1900s.
The most universally supplied agent is
chlorine. Chlorine
combines with water to form hypochlorous acid,
a highly efficient bacteriocide.
The amount of hypochlorous acid
formed by a dose of a chlorine compound will depend on the
amount of organic material and ammonia present, and the pH
of the
water. Typical
chlorine amounts in the range of 1.0 mg/1 will
provide adequate protection for fairly clear water; however,
suspended solids can protect pathogens from the disinfectant
and
result in incomplete disinfection.
Thus, any water that is
disinfected by chlorine should be free of high levels of
suspended
solids.
One of the major advantages of the halogen disinfectants is
their
ability to form stable residuals, which continue to protect
the
water from recontamination.
Depending on the quality of the
water, the residual can persist for as long as a week in the
absence of light.
(The chlorine residual is quickly reduced in
the presence of sunlight.)
One major disadvantage of the residual,
however, is the possibility that the water will develop a
medicinal or chlorinous taste and odor.
The foul taste and odor
are usually not caused by the chlorine (or any other
halogen),
but by compounds that have formed with the chlorine.
A common
contaminant, phenol, yields a strong, distinct odor that is
detectable at very low levels.
In certain situations, chlorinous
odors can be removed by increasing the chlorine dosage,
which
oxidizes the odor-causing compounds.
In the absence of a sophisticated
laboratory, the suitable amount of chemicals needed for
this purpose can be determined by trial and error.
Table 1
provides instructions for chlorinating drinking water.
Many techniques are available for putting the chemicals into
water, ranging from a single dose into a container to a
continuous
feed from some type of storage vessel.
In considering a
technique for small-scale use, the reliability and ease of
use
should be given very high regard.
Any technique that is not used
correctly could yield a false sense of security that could
be
quite dangerous.
Boiling
Boiling is perhaps the most well-known and universally
applied
method of disinfection.
The common consumption of boiled drinks
(teas) was undoubtedly fostered by the realization that
these
drinks were "healthful" (or, more appropriately,
non-pathogenic).
Boiling water--even turbid water--for three to five minutes
effectively destroys all pathogens.
However, boiled water often
tastes "flat."
This flat taste can be remedied by allowing the
water to stand for one or more days while exposing it to the
air.
Typically, 1 kilogram of wood is required to boil about 1
liter
of water.
Caution should be exercised in storing boiled water, as the
potential for recontamination is quite high.
The water should be
stored in a closed, dark container, preferably in a cool
location.
As with any stored water, care should be taken to avoid
contaminating the water when taking water out of the
container.
III. SUMMARY
In developing a treatment system for a small water supply,
emphasis
should be placed first on securing the highest quality of
water possible (e.g., rain water, ground water, surface
water).
Beyond this, any treatment technique that is readily available,
affordable, simple to maintain and operate, and capable of
improving
the quality of the water can be used.
In some cases, it
may be impossible to provide chlorination due to the
unavailability
of raw material or the unreliability of operation.
Other forms of treatment, although less efficient than
chlorination,
may be more reliable and thus provide a consistently better
quality of water than would a less reliable treatment
technique.
The most effective treatment technique is one that will not yield
water if it is not operating properly.
To some extent, filtration
systems meet this criterion and thus are very attractive as
a reliable, small-scale form of treatment.
Additional disinfection
is always desirable, however, to ensure pathogen-free drinking
water.
Table 1.
Amounts of Chemicals Needed to Disinfect
Water for Drinking [a]
Water
Bleaching Powder
High Strength
Liquid Bleach
(m3) (25-35%)
(g) Cal-Hypochl
(52% sodium
(70%)
(g) hypochlorite (ml)
1
2.3
1
14
1.2
3
1.2
17
1.5
3.5
1.5
21
2
5
2 28
2.5
6
2.5
35
3
7
3
42
4
9
4
56
5
12
5
70
6
14
6
84
7
16
7
98
8
19
8
110
10
23
10
140
12
28
12
170
15
35
15
210
20
50
20
280
30
70
30
420
40
90
40
560
50
120
50
700
60
140
60
840
70
160
70
980
80
190
80 1,100
100 230
100
1,400
120 280
120
1,700
150 350
150
2,100
200 470
200
2,800
250 580
250
3,500
300 700
300
4,200
400 940
400
5,600
500 1,170
500
7,000
[a] Approximate dose
= 0.7 mg of applied chlorine
per liter of
water.
Note: For
chlorinating drinking water, follow these instructions:
(1) use one of
the chemicals listed in the table, and
choose the
amount according to the quantity of water in the
distribution
tank, cistern, or tanker; (2) dissolve the
chemicals
first in a bucket of water (not more than about
100 g of
calcium hypochlorite or bleaching powder in one
bucket of
water), and pour the solution into the tank (if
possible,
agitate the water to ensure good mixing); and (3)
repeat this
chlorination procedure as soon as the level of
residual
chlorine in the water drops below 0.2 mg per liter.
Source: S. Rajagopalan.
Guide to Simple Sanitary for
the Control
of Enteric Diseases, (Geneva, World Health
Organization, 1974.)
BIBLIOGRAPHY
Baker, M.N. Quest for Pure Water.
Denver, Colorado:
American
Water Works Association,
1949.
Mazariegos, J. Fernando, and de Zeissig, Julia Alicia
A. Water
Purification
Using Small Artisan Filters.
Guatemala: Central
American
Research Institute for Industry, 1981.
Swiss Association for Technical Assistance, ed.
Manual for Rural
Water
Supply. Zurich, Switzerland:
Swiss Center for Appropriate
Technology,
1980.
United Nations.
World Health Organization.
"WHO Guidelines for
Drinking Water
Quality," by H.G. Gorchev and G. Ozolins.
Geneva, Switzerland:
World Health organization, 1982.
United Nations.
World Health Organization. WHO
International
Reference Centre
for Community Water Supply. The
Purification
of Water on a
Small Scale." WHO Technical Paper
no. 3,
March 1973.
United Nations.
World Health Organization. WHO
International
Reference Centre
for Community Water Supply.
"Preliminary
List of
References on Slow Sand Filtration and Related
Simple
Pretreatment Methods." The Hague,
The Netherlands:
WHO International Reference Centre for
Community Water Supply,
July 1976.
U.S. Agency for International Development.
Development Information
Center.
"Methods of Water Treatment."
Technical Note
no. RWS. 3.M
Water for the World.
U.S. Agency for International Development.
Development Information
Center.
"Determining the Need for Water
Treatment."
Technical Note
no. RWS 3.P.1. Water for the World.
U.S. Agency for International Development.
Development Information
Center.
"Planning a Water Treatment
System." Technical
Note no. RWS
3.P:4. Water for the World.
U.S. Agency for International Development.
Development Information
Center.
"Designing a Small Community
Sedimentation
Basin." Technical Note no.
RWS. 3.D.2. Water for the
World.
U.S. Agency for International Development.
Development Information
Center.
"Constructing a Sedimentation
Basin." Technical
Note no.
RWS. 3.C.2. Water for the World.
U.S. Agency for International Development.
Development Information
Center.
"Designing a Slow Sand Filter."
Technical Note
no. RWS. 3.D.3.
Water for the World.
U.S. Agency for International Development.
Development Information
Center.
"Constructing a Slow Sand Filter."
Technical
no. RWS. 3.C.3.
Water for the World.
U.S. Agency for International Development. Development
Information
Center.
"Operating and Maintaining Slow Sand
Filters."
Technical Note
no. RWS. 3.0.3. Water for the World.
========================================
========================================

