TECHNICAL PAPER # 3
UNDERSTANDING ETHANOL FUEL
PRODUCTION AND USE
By
Cliff Bradley & Ken Runnion
Technical Reviewers
Kenneth Brunot
C. Gene Haugh
Daniel Ingold
Published By
VITA
1600 Wilson Boulevard, Suite 500
Arlington, Virginia 22209 USA
Tel: 703/276-1800 . Fax: 703/243-1865
Internet: pr-info@vita.org
Understanding Ethanol Fuel Production
and Use
ISBN: 0-86619-203-4
[C]1984, Volunteers in Technical Assistance
PREFACE
This paper is one of a series published by Volunteers in
Technical
Assistance to provide an introduction to specific
state-of-the-art
technologies of interest to people in developing countries.
The papers are intended to be used as guidelines to help
people choose technologies that are suitable to their
situations.
They are not intended to provide construction or
implementation
details. People are
urged to contact VITA or a similar organization
for further information and technical assistance if they
find that a particular technology seems to meet their needs.
The papers in the series were written, reviewed, and
illustrated
almost entirely by VITA Volunteer technical experts on a
purely
voluntary basis.
Some 500 volunteers were involved in the production
of the first 100 titles issued, contributing approximately
5,000 hours of their time.
VITA staff included Leslie Gottschalk
as primary editor, Julie Berman handling typesetting and
layout,
and Margaret Crouch as project manager.
Cliff Bradley and Ken Runnion, co-authors of this paper,
specialize in alcohol fuel production technologies.
Bradley is a
microbiologist and Runnion a chemical engineer at Renewable
Technologies, Inc.
They have published several practical manuals
and booklets in the field, and are currently researching and
developing new methods of improving the starch hydrolysis
process
required for alcohol fuel production.
Reviewers Kenneth Brunot,
C. Gene Haugh, and Daniel Ingold are also specialists in the
area. Brunot, senior
vice president for Wright Technology, was
formerly president of Wright Energy Nevada Corporation,
where he
specialized in studies relating to ethanol production using
geothermal energy for process energy requirements.
Haugh heads
the Department of Agricultural Engineering at Virginia
Polytechnic
Institute. Ingold is
a biophysicist by training and a
research engineer at Appropriate Technology Corporation.
VITA is a private, nonprofit organization that supports
people
working on technical problems in developing countries.
VITA offers
information and assistance aimed at helping individuals and
groups to select and implement technologies appropriate to
their
situations. VITA
maintains an international Inquiry Service, a
specialized documentation center, and a computerized roster
of
volunteer technical consultants; manages long-term field
projects;
and publishes a variety of technical manuals and papers.
UNDERSTANDING ETHANOL FUEL PRODUCTION AND USE
By
VITA Volunteers Cliff Bradley and Ken Runnion
I. INTRODUCTION
This paper describes the production and use of ethanol
(ethyl alcohol)
as a liquid fuel.
The production of ethanol is a well-established
technology; however, the use of ethanol as a liquid
fuel is a complex subject.
Ethanol was one of the first fuels used in automobile
engines. It
was used extensively in Germany during World War II and also
in
Brazil, the Philippines, and the United States.
During the postwar
period, as petroleum supplies became cheap and abundant,
gasoline largely replaced ethanol as an automotive
fuel. Not
until the 1970s, when the supply of oil was restricted, did
ethanol re-emerge as an alternative to or extender for
petroleum-based
liquid fuels (ethanol as an extender is added to these
fuels to increase their volume).
Today, 12 countries produce and
use a significant amount of ethanol.
In Brazil, for example, one
third of that country's automobiles uses pure ethanol as
fuel;
the remaining two thirds use mixtures of gasoline and
ethanol.
France, the United States, Indonesia, the Philippines,
Guatemala,
Costa Pica, Argentina, the Republic of South Africa, Kenya,
Thailand, and Sudan are other countries with government or
private
ethanol fuel programs.
The programs are designed to reduce a
country's dependence on costly imported fuel and to assist
in
creating a new domestic fuel industry.
Pure ethanol can replace gasoline in modified spark-ignition
engines, or it can be blended with gasoline at up to 20
percent
concentration to fuel unmodified gasoline engines.
Blending
serves two purposes: (1) it extends gasoline supplies, and
(2)
as an octane enhancer, it replaces lead compounds in
gasoline.
Ethanol can also be used in modified diesel (compression
ignition)
engines; however, this is not common.
The production and use of fuel ethanol can indirectly serve
a variety
of needs. On a
national level, ethanol can improve balance
of payments by displacing imported petroleum with
domestically
produced fuel. This
may also provide increased rural employment
and alternative markets for agricultural commodities.
On a community
or individual level, ethanol fuel production is often
viewed as a means to become independent of purchased fuels,
to
keep money within the local economy, and to provide an
assured
fuel supply in the event of shortages of petroleum fuels.
II. OPERATING PRINCIPLES
ETHANOL PRODUCTION
Ethanol fuel production is a combination of biological and
physical
processes. Ethanol
is produced by fermentation of sugars with
yeast. It is
concentrated to fuel grade by distillation.
Figure 1
uef1x3.gif (600x600)
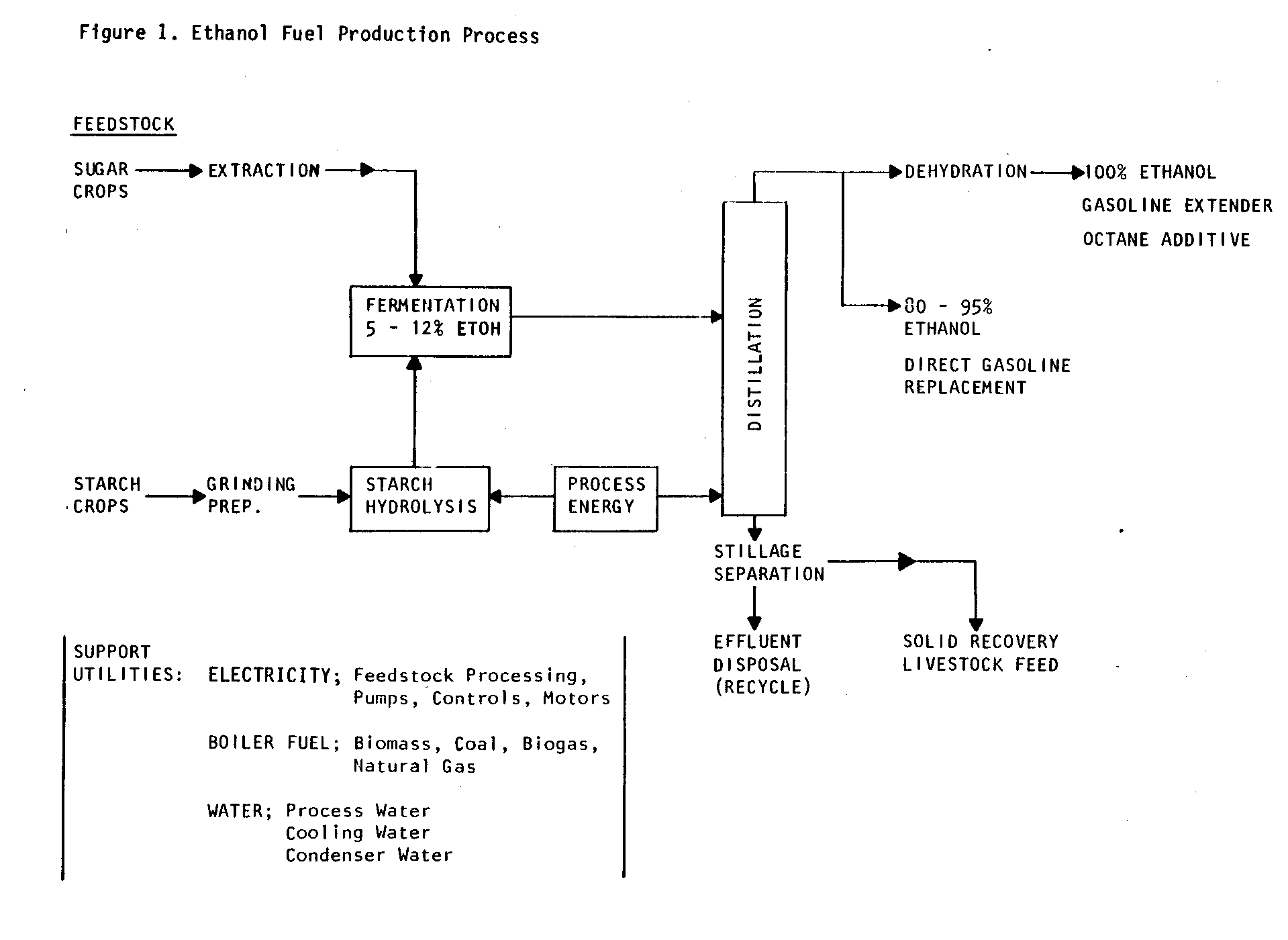
is a schematic representation of the principal steps in fuel
ethanol production.
Feedstocks, the base raw materials, are either sugar or
starch-containing
crops. These
"Biomass Fuel Crops" (tubers and grains)
commonly include sugar beets, potatoes, corn, wheat, barley,
Jerusalem artichokes, and sweet sorghum.
Sugar crops such as
sugar cane, sugar beets, or sweet sorghum are extracted to
produce
a sugar-containing solution that can be directly fermented
by yeast. Starch
feedstocks, however, must be carried through an
additional conversion step.
Starch is a long "chain" polymer of glucose (i.e.,
many glucose
polymer units bonded in a chain).
Starches cannot be directly
fermented to ethanol.
They must first be broken down into the
simpler glucose units through a process of hydrolysis.
In the
hydrolysis step, starch feedstocks are ground and mixed with
water to produce a mash typically containing 15 to 20
percent
starch. The mash is
then cooked at boiling point or above and
treated in sequence with two enzyme preparations.
The first
enzyme hydrolyzes starch molecules to short chains; the
second
enzyme hydrolyzes the short chains to glucose.
The mash is then
cooled to 30[degrees] C, and yeast is added.
Yeasts are microorganisms that produce ethanol.
These microorganisms
are capable of converting sugar into alcohol by a biological
process called fermentation.
The following equation shows the
uefxa2.gif (486x486)

basic biological reaction in the conversion by fermentation
of
one kilogram of glucose to ethanol, carbon dioxide, and
heat:
Theoretically, the maximum conversion efficiency of glucose
to
ethanol is 51 percent on a weight basis.
However, some glucose is
used by the yeast for the production of cell mass and for
metabolic
products other than ethanol.
In practice, between 40 and 48
percent of glucose is converted to ethanol.
With a 45-percent
fermentation efficiency, 1,000 kilograms of fermentable
sugar
produce about 570 liters of pure ethanol.
Conversely, about 1,800
kilograms of fermentable sugar are required to produce 1,000
liters of ethanol.
Mash typically contains between 50 and 100
grams of ethanol per liter (5 to 10 percent weight per
volume)
when fermentation is complete.
Ethanol is separated from mash by distillation--a physical
process
in which the components of a solution (in this case, water
and ethanol) are separated by differences in boiling point
or
vapor pressure.
Ethanol and water form an azeotrope, or constant boiling
solution,
of about 95 percent alcohol and five percent water.
The
five percent water cannot be separated by conventional
distillation.
The production of pure, water-free (anhydrous) ethanol
requires a dehydration step following distillation.
Dehydration,
a relatively complex step in ethanol fuel production, is
accomplished
in one of two ways.
The first method uses a third
liquid, most commonly benzene, which is added to the
ethanol/
water mixture. This
changes the boiling characteristics of the
solution, allowing separation of anhydrous ethanol.
The second
method employs molecular sieves that selectively absorb
water on
the basis of the difference in molecular size between water
and
ethanol.
The non-fermentable solids in distilled mash (stillage)
contain
variable amounts of fiber and protein, depending on the
feedstock.
The liquid may also contain soluble protein and other
nutrients.
The recovery of the protein and other nutrients in stillage
for use as livestock feed can be essential for economical
ethanol fuel production.
Protein content will vary with feedstock.
Some grains (e.g., corn, barley) yield a solid by-product
--distillers dried grains (DDG)--that ranges from 25 to 30
percent
protein and makes an excellent feed for livestock.
If the
processing equipment is constructed of stainless steel and
processing
is carried out under well-controlled conditions, the protein
by-products can also be consumed by humans.
The production of ethanol also produces liquid effluent,
which is
a potential pollution problem.
About 9 liters of effluent are
produced for each liter of ethanol.
In well-designed plants, some
of the effluent may be recycled.
Effluent can have a high Biological
Oxygen Demand (BOD), which is a measure of organic water
pollution potential, and it is acidic.
It requires treatment
before discharge.
Treatment requirements depend on feedstock and
local pollution control regulations.
Because of the acid content,
care must be taken if the effluent is spread over fields.
ETHANOL END USE
Ethanol is a high-quality, stable liquid.
Some of the chemical
and physical properties of ethanol are summarized in Table
1.
Table 1. Summary of Ethanol
Properties
Property
Ethanol
Chemical Formula..............................
[C.sub.2][H.sub.5]OH
Molecular Weight..............................
46.07
Density (20[degrees] C).......................
0.791 g/cc
Boiling Point [a].............................
78.5[degrees] C
Heat of Combustion [b]........................
5625 Kcal/1
Heat of Vaporization [c]......................
9.225 Kcal/mole
Octane Rating.................................
106-108
Stoichiometric Air/Fuel Ratio [d].............
9/1
[a] Boiling point is
the temperature at which a liquid changes
phase and
becomes a gas; the point at which the vapor pressure
of the liquid
equals the vapor pressure of the system.
[b] Heat of
combustion is the amount of heat given off when a
unit quantity of
any hydrocarbon (e.g., ethanol) is burned
to carbon
dioxide and water.
[c] Heat of
vaporization is the heat input required to change
liquid at its
boiling point to a vapor at the same temperature
(e.g., water at
100[degrees] C to steam at 100[degrees] C).
[d] The
stoichiometric air/fuel ratio is the amount of air
necessary
completely to oxidize (burn) the fuel.
Ethanol Use in Engines
Ethanol is most commonly used in transportation and
agriculture
to fuel internal combustion, four-cycle, spark-ignition
engines.
It is used as a direct replacement for gasoline, or it is
blended
with gasoline as an extender and octane booster.
The use of ethanol to replace gasoline requires
modifications to
the carburetor, fuel system components, and often the
compression
ratio. The efficient
conversion of existing gasoline engines requires
skilled, knowledgeable technicians.
Engines specifically designed and manufactured to operate on
ethanol fuel will generally be more efficient than modified
gasoline
engines. Ethanol
concentrations of between 80 and 95 percent
can be used as fuel, which eliminates the need for
sophisticated
dehydration systems and simplifies distillation.
In many cases,
the conversion of engines to operate on ethanol may be
simpler
and more cost efficient than ethanol dehydration.
The disadvantage
of engine conversion is that vehicle travel distance is
limited
by the available supply and distribution of ethanol.
Some "dual fuel" systems--that is, engines with a
carburetor that
can operate either on ethanol or on gasoline--have been
developed
on a limited basis.
In Brazil, a significant portion of the
transportation fleet uses ethanol fuel in automobiles with
specially
designed engines, manufactured by major international
automobile
companies.
In unmodified engines, ethanol can replace up to 20 percent
of
the gasoline.
Blending ethanol with gasoline extends the gasoline
supply, and improves the quality of gasoline by increasing
its
octane value. As an
octane enhancer, ethanol can replace lead
compounds in gasoline.
There are advantages to using gasoline/
ethanol blends rather than pure ethanol.
Blends do not require
engine modification.
In this way, ethanol can be integrated
rapidly with existing gasoline supply and distribution
systems.
Replacing lead compounds with ethanol removes one of the
principal
air pollution problems associated with gasoline.
The disadvantage of using ethanol/gasoline blends is that
the
ethanol must be anhydrous, requiring a dehydration step in
production.
If non-anhydrous ethanol is mixed with gasoline, the
blends will separate into a gasoline phase and a
water/ethanol
phase, causing erratic engine performance.
In addition to its use in gasoline-fueled automobiles and in
truck or tractor engines, ethanol can be used in other types
of
engines. For
example, small, four-cycle gasoline engines found in
small-scale agricultural equipment (e.g., tillers, small
tractors)
can often burn 80 to 95 percent ethanol as a direct
replacement
for gasoline. Such
engines fed by ethanol require
minimal modifications.
The use of ethanol in specially designed two-cycle engines
has
been demonstrated on a limited basis.
The problem of using ethanol
in these engines is that the ethanol does not blend well
with
lubricating oil. To
get around this problem, research is under
way to find lubricating oils that are not affected by
ethanol.
Though ethanol use in diesel-fueled engines is feasible, it
has
its limitations.
Ethanol does not ignite under compression and
does not mix well with diesel fuel.
Therefore, ethanol cannot be
used as a direct replacement for diesel fuel or blended with
diesel fuel for use in compression ignition engines.
Ethanol can
be used as a replacement for diesel fuel only if the engine
is
fitted with glow plugs.
Ethanol can be used in supercharged diesel engines for up to
about 25 percent of the total fuel.
This is done by carrying the
ethanol in a separate fuel tank and injecting it into the
diesel
engine through a supercharger airstream.
Ethanol can also replace aviation fuel in aircraft engines.
Ethanol Use in Appliances
Ethanol can be used in a variety of cooking, heating, and
lighting
appliances. In some
cases, ethanol can be used in modified
appliances designed for conventional fuels.
In other cases,
appliances designed specifically for ethanol fuel are
required.
III. PLANT DESIGN VARIATIONS
This section describes briefly the processes and equipment
necessary
for each principal step in ethanol fuel production.
It also
provides a general discussion of the economics of ethanol
fuel
production. It is
not meant to provide specific information on
plant design.
Processes and equipment vary greatly, depending on
feedstock, the
need for starch hydrolysis, ethanol end use, available
support
utilities, process energy source, by-product use, and plant
scale.
FEEDSTOCK PROCESSING
Plant design studies indicate that an economy of scale
exists for
a 30,000,000 gal/year plant producing hydrated (190 proof)
ethanol
and co-generating, i.e., utilizing on-site gas turbine
generator
sets fueled with hydrated ethanol to provide associated
power needs for the plant.
The turbine exhaust gas could be used
to obtain high-pressure steam and the spent hot turbine
exhaust
gas could be used in process by-product drying
operations. Provision
to produce process by-products (distillers dried grains
(DDG), carbon dioxide, and fusel oil components should be
included
in the overall design in order to maximize cost
effectiveness.
The type of feedstock chosen for ethanol fuel production has
a
significant impact on plant design.
Ethanol is produced from a
variety of sugar- or starch-containing crops, with
modifications
in the design of the feedstock preparation processes.
The
modifications are required to accommodate the physical
properties
of the feedstock, as well as the nature of the carbohydrate
(i.e., sugar versus starch).
Preparation equipment is necessary to grind, pulverizer or
extract
the feedstock before it can be processed.
Milling equipment
for feedstock preparation varies, depending on such
characteristics
of the feedstock as moisture content, physical structure,
and fiber content.
Starch Hydrolysis
Starch-containing feedstocks require starch hydrolysis
equipment
including tanks, heating and cooling systems, agitation
systems,
transfer pumps, and monitoring instruments.
Starchy feedstocks
have to be ground before hydrolysis to a particle size that
can
pass through a 20-mesh screen.
Steam circulated through heat exchangers is the most common
means
of heating the mash; therefore, starch hydrolysis heating
requirements
must be included in plant boiler capacity.
Cooling the mash from boiling to fermentation temperature
(about 30[degrees] C) generally the determining factor in
heat exchanger
design. This is
especially true in tropical climates
where the ambient temperature of the cooling water is
relatively
high.
The agitation systems for starch hydrolysis tanks must be
adequate
to mix viscous (thick) starch solutions efficiently.
When
starch is heated in water, it forms a very thick gel.
Starch
gelatinization is essential for efficient enzymatic
hydrolysis.
Thorough mixing of gelled starch mash is necessary to ensure
efficient heat exchange and enzyme activity.
Monitoring equipment for starch hydrolysis includes
thermometers
to measure mash temperature and steam temperature, and
pressure
gauges to measure mash pressure if pressurized starch
hydrolysis
systems are used.
Tests to measure the efficiency of starch hydrolysis
are also necessary.
Generally speaking, the feedstock is
the most important element in determining the economics of
ethanol
production, and inefficient starch hydrolysis can have a
major economic impact on ethanol production.
Starch hydrolysis systems are of two general types: batch
systems
and continuous systems.
Batch systems consist of tanks that are
sized in relation to fermentation tank capacity and holding
time.
The tank is equipped with heat exchangers, usually internal
coils, that circulate steam and cooling water.
The mash is
agitated by a motor equipped with gear reduction and mixing
impellers. Transfer
pumps capable of handling a high level of
solids are used to transfer the mash of fermentation
tanks. With
very viscous feedstocks, heat exchange and mash agitation
are
accomplished by pumping the mash through an external heat
exchanger
and back into the tank.
Batch systems are operated by
filling the tank, carrying out the multistep process of
enzyme
hydrolysis, and then pumping the entire mash volume into
fermenters.
Continuous starch hydrolysis systems require more
sophisticated
equipment, but they are usually more efficient.
Continuous systems
generally use "jet cookers," in which mash and
stem are
mixed under pressure at temperatures of 105 to 150[degrees]
C. Water,
feedstock, and enzyme are fed into a premix tank at a
controlled
rate, heated, and pumped under pressure through the
jet. The mash
is kept at high pressure and high temperatures for a few
minutes,
then released from the cooker to holding tanks, where it is
cooled and additional enzyme is added.
The mash is then transferred
to fermenters. The
high pressure and temperature of these
systems result in more efficient starch gelatinization and
hydrolysis.
These systems require high-pressure boilers and relatively
sophisticated systems for maintaining the mash under
pressure.
Equipment is sized on the basis of plant fermenter capacity
and mash residence time in cookers.
Fermentation
Fermentation takes place in tanks equipped with agitation
and
heat exchangers to remove the heat generated by fermentation.
Tank size is based on the concentration of sugar in the
mash,
fermentation time, final ethanol concentration, and plant
production
rate.
Final mash ethanol concentration is a direct function of
mash
sugar concentration.
Within the limits of feedstocks and yeast
ethanol tolerance, higher ethanol concentrations are
desirable.
Maximum mash ethanol concentration is about 10 percent
weight per
volume. At
concentrations higher than 10 percent, yeast are
killed. Generally,
feedstocks with high moisture content and
sugar or starch concentrations less than 20 percent can be
fermented
without dilution.
Feedstocks with high starch or sugar
concentrations require dilution.
The sugar will be wasted if the
concentration is in excess of the amount necessary to
produce the
maximum amount of ethanol tolerated by the yeast.
Fermentation typically requires from 12 to 72 hours
depending on
the amount of yeast used to start fermentation and mash
sugar
concentration.
Plants are usually equipped with multiple fermentation
tanks run on staggered schedules to provide a continuous
supply of fermented mash for distillation.
One of the most significant problems in ethanol production,
especially in small-scale plants, is contamination of mash
by
bacteria. Bacteria
utilize sugars that would otherwise be converted
to ethanol. Good
plant design and efficient fermentation
can control contamination wihout resorting to costly
sterilization
systems.
Distillation
Distillation systems can either be batch or continuous.
Choosing
one system or the other is based on plant scale.
Both types
require heating systems, usually steam (which can be from
low-pressure
boilers), a distillation column, and a condenser.
Figure 2 shows schematics of these two types of systems.
uef2x110.gif (600x600)

Distillation column size and ethanol production rate are
based on
the concentration of ethanol in the fermented mash,
fermentation
capacity, and production schedules.
Small-scale plants--up to
about 100,000 liters annual ethanol production--can
efficiently
use batch distillation systems.
In batch systems, the entire mash
volume is passed, or charged, to a large vessel called a
still,
which is then heated.
The vapors are allowed to pass into the
distillation column.
Though batch systems are less efficient than
continuous feed distillation systems, they are much easier
to
build and operate.
In continuous feed systems, fermented mash is pumped at a
controlled
rate into the distillation column, with heat introduced
at the bottom of the column.
Provision is made at the top of
the column to feed unprocessed mash back through the system.
Continuous feed columns should be used in large-scale plants
where the improved efficiency justifies the added
complexity.
Dehydration
The intended use of the ethanol determines the need for
dehydration
systems to remove the five percent water that cannot be
separated by distillation.
If ethanol is to be blended with
gasoline, dehydration is required.
The presence of water in
ethanol gasoline-blends results in phase separation in
storage
or fuel tanks.
Dehydration is not required if ethanol is to be
used to replace gasoline.
Ethanol can be used directly in modified
engines at concentrations of between 80 and 95 percent.
By-product Recovery
Solid by-products are recovered from stillage with
solid/liquid
separation equipment.
This equipment can range from simple
screens to such complex equipment as centrifuges or vacuum
filters.
Soluble protein in thin stillage can be recovered by
evaporation. If
by-products are to be stored or transported
significant distances, drying is necessary.
Stillage with high
moisture content can often be fed directly to livestock at
or
near the site of ethanol production with minimal separation
or
processing.
Effluent Treatment
Production of each volume of fuel ethanol will generate
about
nine volumes of effluent.
A portion of the effluent can be recycled
and used to dilute high concentration feedstocks.
However,
even if the effluent is recycled, it can still cause a
significant
pollution problem.
To avoid pollution of surface water or
ground water, the effluent must undergo microbiological
degradation;
that is, the harmful organic matter contained in the
effluent must be broken down before the effluent is disposed
of.
This is done anaerobically, aerobically, or by a sequential
combination of the two methods.
Effluent degradation is usually
done in a simple treatment pond, followed by a stabilization
pond, if necessary.
Alternatively, the effluent can be fed to
biogas digesters, combining energy production with waste
treatment.
Utilities
Ethanol production requires water, boiler fuel, and
transportation
for feedstock, ethanol, and by-products.
Electricity may be
used to run pumps, stirring motors, process controls, and
instrumentation,
but there are many units throughout the world that
produce up to 10,000 gallons per year without
electricity. Water
is required for mash dilution and cooling in heat exchangers
used
with starch hydrolysis systems, fermenters, and condensers.
Boilers used to generate process steam require a low-cost,
low-quality
fuel such as natural gas, biogas, biomass, coal, residual
oil, or bagasse (bagasse is crushed sugar cane or beet
refuse
from sugar making).
High-quality liquid fuels or electricity are
uneconomical and inefficient for use as boiler fuel.
Finally, the
feedstock needs to be transported to the plant; and the
products,
both ethanol and by-product animal feed, need to be
transported
to the point of use.
The process energy requirements for ethanol fuel production
vary
widely depending on equipment, process design, and
feedstock. The
production of one liter of ethanol with a heating value of
5,625
Kcal/liter would typically require 800 to 1,200 Kcal for
cooking,
1,300 to 1,500 Kcal for distillation, 800 to 1,000 Kcal for
dehydration, and about 300 Kcal to operate stirring motors
and
pumps. The drying of
by-products from the production of one
liter of ethanol might require an additional 600 to 700
Kcal. The
production of anhydrous ethanol and dried by-products from
grain--representing the high range for process energy--would
require 2,800 to 3,800 Kcal/liter.
The production of 90 percent
ethanol from sugar feedstock without by-product
drying--representing
the low range for process energy--would require 1,600 to
1,800 Kcal/liter.
The availability and the cost of utilities are critical
factors
both in the scale and the economics of alcohol
production. Two
factors have contributed to the failure of ethanol fuel
projects
in developing countries:
(1) plants for the production of ethanol
fuel were so large that the support utilities were unable to
meet
their energy needs; and (2) the plants were sited so far
from the
feedstock that transportation was not economical.
Plant Scale
Ethanol fuel plants range in size from a few thousand liters
to
more than 100 million liters of annual production.
The design and
operation of small-scale plants can be greatly simplified by
combining starch hydrolysis, fermentation, and batch
distillation
in multi-purpose process tanks.
The plants could include one or
several tanks supplying fermented mash for a single
distillation
column. Plants up to
about 100,000 liters of annual production
can be designed this way.
Even larger batch plants can be considered
if low-cost boiler fuel is available.
With good technical
assistance, small-scale batch plants can be built and
operated
with local, community-level resources and skills.
Greater operating efficiency can be obtained in larger
plants by
separating starch hydrolysis and fermentation in specially
designed
systems and using continuous feed distillation columns.
Generally, the greater capital cost and operating complexity
of
this type of plant will be returned in operating efficiency.
Large-scale alcohol plants require at least some employees
with
relatively sophisticated management and technical
skills. Plant
design, equipment, and construction often require resources
outside
the local community.
COST/ECONOMICS
It is difficult to provide general information about ethanol
fuel
economics because production costs and product value depend
on
plant location, feedstock, production scale, and end use.
Ethanol production includes both capital and operating
costs. Two
important factors in capital costs for small batch plants are
starch hydrolysis systems and boiler capacity.
In large plants,
engineering, distillation systems, and process control are
relatively
more significant.
Generally, capital costs for alcohol
plants range from $.50 to $1 (U.S.) per liter of annual
production
capacity. Based on
figures from U.S. plants, capital
costs per liter of annual production capacity for very small
and
very large plants are generally greater than those for
intermediate-scale
plants--1 to 10 million liters annual production.
The greatest operating cost in ethanol production,
regardless of
scale, is feedstock.
For ethanol fuel production to be profitable,
an economical supply of feedstock is essential.
In small
plants, labor-costs may also be relatively important.
Indirect costs for engine conversion, distribution and
marketing,
plant utilities, and transportation of feedstock and
products are
also important in evaluating ethanol production costs.
Ethanol market value depends on end use.
The market value of
ethanol as a replacement fuel would generally be measured
relative
to gasoline prices.
The market value of ethanol when blended
with gasoline may be higher than gasoline because of the
increased octane value of ethanol/gasoline blends.
By-product market value is measured against the local price
of
animal feed. The
value is typically determined by comparing the
protein content of feeds.
Other factors, aside from ethanol production costs and the
market
value of ethanol, may also be significant to the economic
analysis.
Displacement of imported petroleum with domestically
produced
renewable fuel may improve balance-of-payment deficits and
may be economically advantageous despite relatively higher
ethanol
costs. Opportunities
for rural employment, alternative markets
for agricultural commodities, and energy independence may
provide significant economic advantages in addition to a
direct
accounting of plant profitability.
IV. COMPARING THE
ALTERNATIVES
FUELS COMPETING WITH ETHANOL
Methanol, butanol, and some types of vegetable oil are three
alternatives
to ethanol. Both
methanol and butanol can be used to
replace or extend gasoline or diesel fuel.
Vegetable oils, however,
are limited to replacing only diesel fuel until further
research
proves otherwise.
Methanol is the most important alternative.
It is a liquid alcohol
containing one carbon atom ([CH.sub.3]OH).
Like ethanol, it is used
to replace or be blended with gasoline.
Methanol is produced by a
chemical process that uses methane as the primary feedstock.
Methanol can also be produced from coal or biomass.
On a
worldwide scale, the methanol production industry is
relatively
large, and it uses natural gas for feedstock.
Methanol production
requires high temperature, high pressure, and special
catalysts.
This process is much more complex than ethanol production
and is
generally economical in only very large industrial plants.
Butanol is a four-carbon alcohol.
It has two possible chemical
structures, depending on the position of the hydroxyl:
N butanol
([CH.sub.3] [CH.sub.2] [CH.sub.2][CH.sub.2]OH) and 2 butanol
([CH.sub.3] [OH.sub.1] [CH.sub.2] [CH.sub.3]).
Fermentation
produces N butanol.
Unlike ethanol or methanol, butanol can
substitute for or be blended with diesel fuel in compression
ignition engines. It
is produced by bacterial fermentation of
starch- or sugar-containing feedstocks and purified by
distillation.
The bacteria produce ethanol and acetone in addition to the
principal product, butanol.
The production of butanol has two disadvantages:
(1) the fermentation
of butanol is difficult compared with that of ethanol; and
(2) butanol fermentation produces less-useful fuel per unit
of
feedstock than ethanol fermentation with yeast.
Butanol has been
produced commercially under wartime conditions.
Today, however,
butanol is no longer produced commercially for use as fuel.
CURRENT RESEARCH AND DEVELOPMENT
Ethanol fuel production is a well-established commercial
technology.
But it is also a technology that has room to improve.
That is why research and development efforts in ethanol fuel
production are ongoing.
The research areas relating to this
technology that continue to be addressed include (1)
feedstock;
(2) starch hydrolysis and fermentation process design; (3)
ethanol and by-product end uses; and (4) site-specific
integration
of ethanol production with local agricultural economics.
Feedstock is the most significant cost element in ethanol
production.
Questions of possible competition for prime agricultural
land, and impacts of ethanol production on food supply and
distribution
are crucial to the social and economic success of this
technology. One
important area of research is the identification
of starch- and/or sugar-containing crops that can be grown
on
poor land and that require a minimum amount of cultivation
and
chemical inputs (e.g., fertilizers).
Such feedstocks must be
compatible with the local climatic conditions, the water
resources,
and the soil type.
They should not disrupt the local agricultural
economy. Alternative
feedstocks under evaluation in
various parts of the world include sago palm, bamboo, sweet
potatoes, and honey locust trees.
Once potential crops are identified,
research will be directed toward increasing yields,
adapting crops to specific situations, and developing
cultivation,
harvest, and storage techniques.
Alternative feedstocks will require research to adapt starch
hydrolysis and fermentation equipment and procedures to the
particular
feedstock characteristics and concentration of fermentable
sugars. Fermentation
research might also include the selection
of yeast strains for improved fermentation efficiency.
Improvements
could include increased tolerance to high sugar and
ethanol concentrations, tolerance to high fermentation
temperature,
or adaptation to particular feedstock characteristics.
Research needs in ethanol and by-product end uses could
include
evaluation of technology and economics for uses of ethanol
other
than as a motor fuel; evaluation of conversion techniques
for
specific types of engines; and evaluation of specific
feedstocks
for recovery and use of by-products.
Research on integration of ethanol fuel production with
agricultural
economies could cover a broad range of topics, including
feedstock economics and cultivation, plant and equipment
design
to fit specific local constraints, process fuel sources,
impacts
on employment and income distribution, and effects on
national
balance of payments.
V. INTEGRATION
The successful introduction of ethanol fuel production and
use in
developing countries requires careful planning.
The technology
must be integrated with local economic conditions, available
resources, and potential end use of both the ethanol and its
by-products.
The operating efficiency of large-scale ethanol plants
may be greater than that of small-scale plants.
However, this
efficiency may be of little value if the plant is too large
for
the available feedstock and support utilities or if the
local
economics of food production and distribution are disrupted.
Ethanol plants should be scaled so that demand for feedstock
does
not disrupt distribution systems and markets for
agricultural
commodities. Support
utilities and transportation should be able
to support the scale of ethanol production.
One important, hidden
cost of large-scale ethanol plants is the cost of building
or
upgrading roads, water supply systems, pollution control
systems,
and electricity generating capacity.
The method used to finance
these support systems is an important economic question.
Distillers dried grains (DDG) are the major by-product
commodity
resulting from ethanol production.
This high protein product is
an excellent livestock feed, and feed lots could be located
near
the ethanol plant.
Another extremely important potential use of
this protein-rich material could be as a human food
supplement.
End use of the ethanol and by-products must be on a scale
that
matches production.
Technical resources need to be available for
engine conversions if necessary.
If ethanol is to be blended with
gasoline, marketing and distribution systems for ethanol and
for
ethanol/gasoline blends must be developed in parallel with
the
construction and operation of ethanol fuel plants.
Proper integration can enhance ethanol production economics
and
can be achieved with well-designed small- and medium-scale
plants. Small-scale
plants can often take advantage of low value
or waste feedstocks such as food processing waste or damaged
or
spoiled crops. A
variety of low-cost boiler fuels such as biogas,
waste heat from other industry or power plants, or biomass
can be
used if the plants are scaled to match the resources
available
within economical transport distances.
Dehydration can be eliminated
if ethanol is used in converted engines.
Alternatively, a
number of small ethanol plants can supply 80 to 95 percent
ethanol
to a centralized plant for dehydration and
distribution. By-product
processing can be reduced if the plant is scaled to
supply livestock feed demand in the immediate area of the
plant.
Small-scale plants are much simpler to build and operate
than
large plants. With
technical support, small-scale ethanol plants
can be built and operated using locally available skills and
resources. With the
exception of such equipment as motors, boilers,
and controls, small-scale plants can be built in any
reasonably
well-equipped machine shop, provided that technically sound
plans are available.
Small-scale plants can also be mounted on
flat-bed trailers so they can be moved from site to site.
Starch hydrolysis and ethanol dehydration are the two steps
requiring long-term purchase of materials outside the local
or
even national level.
The production of starch hydrolysis enzymes
and molecular sieves requires relatively sophisticated
technology.
Enzymes and molecular sieves are supplied by a number of
companies. As an
alternative to purchasing these materials, they
can be manufactured in centralized plants for distribution
to
small-scale ethanol plants.
VI. CHOOSING THE
APPROPRIATE TECHNOLOGY
The decision to produce and use ethanol fuel requires
addressing
both direct and indirect technical and economic
questions. These
questions are important on any scale of development ranging
from
an individual local decision to produce on a small scale to
national-level
programs.
Direct technical and economic questions in the decision to
produce
and use ethanol fuel include the cost and the availability
of feedstock; ethanol and by-product end uses and marketing;
laws
and regulations; production scale; and selection of plant
design
and equipment options.
Factors affecting feedstock availability and cost include
transportation,
storage, potential spoilage, and seasonal variations
in supply and price.
Ethanol and by-product uses are affected by product
transportation
and distribution, storage, possible spoilage of by-products,
seasonal variations in market demand or on-site use, and
whether
the ethanol is to replace or be blended with gasoline.
If ethanol
is to be blended with gasoline, the costs and the systems
for
distribution, blending, and marketing need to be taken into
account. If ethanol
is to replace gasoline, the costs of engine
conversion and limitations to vehicle use are two important
factors.
Laws and regulations affecting ethanol fuel production will
vary
from country to country.
Variations may also occur between legal
and political jurisdictions within countries.
Regulations must be
checked for each individual case.
The principal regulations are
those that prevent the use of fuel ethanol for human
consumption.
Generally, these regulations require that ethanol be
denatured by
adding chemical agents to the ethanol to make it unfit for
human
consumption. The
most readily available denaturant for ethanol is
gasoline mixed at one percent per volume.
Other regulations may
govern discharges of liquid and gaseous effluents and
occupational
safety and health.
Laws dictating conformance to building
codes (e.g., electric, plumbing, and fire safety codes) may
also apply.
Decisions regarding plant scale, equipment, and process
design
depend primarily on feedstock, the availability of markets
for
both ethanol and its by-products, and the availability of
plant
financing. Economies
of scale in ethanol fuel production are much
less important than well-planned integration of ethanol fuel
production
with agricultural economics, local transportation, local
utilities, and end uses.
Indirect social and economic questions are also very
important in
the decision to produce and use ethanol fuel.
Economic decisions
regarding ethanol production may rely more on the ability to
meet
such objectives as increasing rural employment, achieving
energy
independence, and providing alternative markets for crops
than on
direct evaluation of production costs and market
values. Technical
decisions regarding plant scale, process design, and
equipment
may be influenced by the ability to meet such objectives as
the use of local labor and locally manufactured equipment,
the
creation of alternative markets for agricultural crops as
feedstocks,
and the local use of process energy.
The emergence of ethanol as a viable alternative to gasoline
has
led to two major controversies that can affect decisions
regarding
ethanol fuel production.
The first controversy concerns the question of net energy
yield;
that is, whether the energy content of the ethanol is
greater
than the energy consumed in production.
With efficient technology,
the energy content of ethanol exceeds the direct in-plant
process energy inputs by about 2 to 1.
However, one recent analysis,
which took into account the energy used to cultivate
feedstocks
and to transport feedstock and products, calculated that
ethanol production consumes more energy than is
produced. The
technical response to this analysis is that ethanol is not a
primary
energy source; rather, it is an energy conversion and
storage system. In
ethanol production, low-quality, diffuse primary
energy sources are upgraded to a high-quality, liquid fuel.
Solar energy in the form of plant carbohydrate and low-quality
boiler fuels is converted to a fuel suitable for use in
transportation.
In simple terms, the response is that automobiles
cannot run on cassava.
When ethanol is viewed as an energy
conversion system, the net energy question is largely
irrelevant.
Nevertheless, the question is useful because it points out
the
need to select those feedstocks requiring relatively little
cultivation and low inputs of fertilizer and chemicals, and
the
need to use low-quality boiler fuels.
The second controversy surrounds the issue of food versus
fuel;
that is, whether the use of agricultural crops for ethanol
fuel
production will adversely affect the amount of land
available for
food production and food supply, as well as affecting food
prices. This is a
complex question to which there are no absolute
answers. On the one
hand, a large-scale diversion of food crops
to ethanol production could reduce food supplies and
increase
food prices. On the
other hand, a carefully planned and well-integrated
ethanol fuel industry does not necessarily result in
direct competition for agricultural land and food
supplies. Low-value
crops grown on marginal land are often good alcohol
feedstocks
with poor food value.
Cultivation of low-value crops may
contribute to the economy through conversion to a high-value
product. Increased
rural employment may increase people's economic
access to high-quality food.
Ethanol might also be produced
from agricultural commodities that would otherwise be
exported.
Sugar cane, for example, may be worth more as a feedstock
for
domestic fuel production to displace imported petroleum than
as
an export crop. The
issue of food versus fuel emphasizes the
need for careful planning but does not mean that ethanol
fuel
production is an inappropriate technology.
BIBLIOGRAPHY
The Bioenergy Council.
The Bioenergy Directory.
Washington,
D.C.:
The Bioenergy Council.
Bernton, Hal; Kovarik, William, and Sklar, Scott.
The Forbidden
Fuel:
Power Alcohol in the Twentieth Century. New
York: Boyd
Griffin, Inc.,
1982.
Brown, Michael H. Brown's Alcohol Motor Fuel Cookbook.
Cornville,
Arizona:
Desert Publications, 1979.
Carley, Larry W. How
To Make Your Own Alcohol Fuels. Blue
Ridge
Summit,
Pennsylvania: Tab Books, Inc., 1980.
Cheremisinoff, Nicholas P.
Gasohol For Energy Production.
Ann
Arbor,
Michigan: Ann Arbor Science Publishers,
1979.
De Rasor, Roberto.
Alcohol Distiller's Manual for Gasohol and
Spirits.
San Antonio, Texas:
Dona Carolina Distillers, 1980.
Development Planning and Research Associates, Inc. Gasohol:
Economic
Feasibility Study 1978. Available from
the National
Technical
Information Service, U.S. Department of Commerce,
5285 Port Royal
Road, Springfield, VA 22161.
First InterAmerican Conference on Renewable Sources of
Energy.
Proceedings of
the First InterAmerican Conference on Renewable
Sources of
Energy, 25-29 November 1979. New
Orleans,
Louisiana:
First InterAmerican Conference on Renewable
Sources of
Energy, 1980.
Hale, William J.
Prosperity Beckons: Dawn of the
Alcohol Era.
Minneapolis,
Minnesota: Rutan Publishing, 1979.
The Mother Earth News.
Making Alcohol Fuel.
Hendersonville, North
Carolina:
The Mother Earth News, 1979.
Solar Energy Information Data Bank, Solar Energy Research
Institute,
U.S. Department
of Energy, Alcohol Fuels Bibliography
(1901 - March
1980). April 1981,
SERI/SP-751-902. This document
is available in
print from the Superintendent of
Documents, U.S.
Government Printing Office, Washington, D.C.
20402, or in
microfiche from the National Technical Information
Service, U.S.
Department of Commerce, 5285 Port Royal
Road,
Springfield, VA 22161.
Solar Energy Information Data Bank, Solar Energy Research
Institute,
U.S. Department
of Energy, Fuel From Farms. A Guide
to Small-Scale
Ethanol Production. 1980.
Also available from
the above
sources.
U.S. Industrial Chemicals Co. Division of National
Distillers and
Chemical
Corporation. Ethyl Alcohol
Handbook. New York, New
York:
U.S. Industrial Chemicals Co., 1969.
Willkie, Herman F., and Kolachov, Paul J. Food For Thought.
Indianapolis,
Indiana: Indiana Farm Bureau, Inc., 1942.
Winston, Paul R. Make Alcohol:
The New Way To Go.
McHenry, Illinois:
For-Wins Inc.
The World Bank.
Emerging Energy and Chemical Applications of
Methanol:
Opportunities For Developing Countries.
Washington,
D. C.:
The World Bank. 1982.
Reference inquiries on specific topics relating to ethanol
fuel
production can be referred through VITA to the staff of
Renewable
Technologies, Inc., who prepared this report, or to other
VITA
volunteers with expertise in ethanol fuel.
========================================
========================================

